Continuing from last week’s blog, we will continue to dive deeper into the fascinating world of biochemistry of fats. The following will build from last week’s blog, so if you haven’t read it, you can read it HERE, or for the summary you can click HERE.
Before I start naming what oils are healthy and unhealthy, you need to understand the process of how the chemical structure of fats break down. By the end, you will know for yourself which oils are safer than others.
Chemistry 101
Sorry to have to do this to you, but a basic understanding of chemistry is needed in order to understand which fats are good or bad for you.
You probably remember from high school chemistry class that atoms contain electrons. These electrons orbit the nucleus of an atom and a certain number of electrons (8 in the outer shell) will make the orbit full and it is then said that the atom is stable. When an atom doesn’t have a full orbit of electrons, it is unstable and will seek out other atoms in order to fill up on electrons. Sometimes, these instances result in happy marriages like hydrogen and oxygen, forming H2O or water.
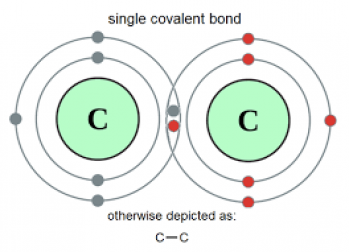
Here is an example of a happy marriage – a single covalent bond where 2 carbon atoms can benefit from sharing their electron:
Image from https://antisensescienceblog.wordpress.com/2013/11/20/
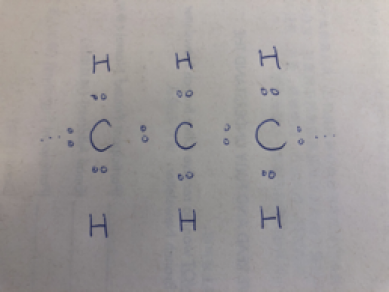
Re-visiting the fatty acid chain,
H is for hydrogen atom
C is for carbon atom
 Figure 1.1
Figure 1.1
In Figure 1.1 above, you can see that the covalent bonds are represented by a “-”. So we can also represent the fatty acid chemical chain like this:
Figure 2
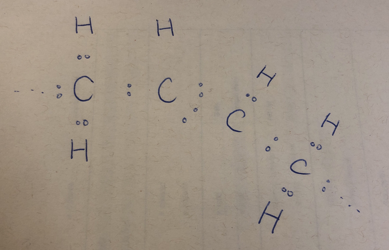
When 2 hydrogen atoms are missing like in a polyunsaturated fatty acids (PUFAs), 2 electrons are also missing:
Figure 3
So the 2 carbon atoms join hands and form not 1, but 2 covalent bonds, hence the name “double bond” (aka. the kink!).
Figure 4
So why am I explaining all this to you?
Without going into the deep technicalities of organic chemistry, the thing you need to understand is that the second bond in a double bond is called a pi bond and is highly unstable.
It is so unstable that any radiation in the form of heat or light has the potential energy to break that pi bond.
When this happens, the electrons that formed the pi bond shards off like broken glass and becomes marauders in the body. They can very easily offset any other atoms or molecules it comes into contact with causing chemical damage known as oxidative stress by free radicals. These free radicals who have now been upset by marauding electrons are now causing further damage with whatever atoms or molecules it encounters and what you have now is a cascading chain reaction of free radicals, one stealing electrons from the next.
If this is left uncheck, oxidative stress that occurs in the skin forms wrinkles and premature aging. If it occurs in the organs, tumors can develop. And if this occurs in the linings of your arteries, then atherosclerosis (aka. plaque formation) can lead to heart disease and stroke.
When you dig into the manufacturing and processing of mainstream cooking oils like soybean, corn, sunflower seed, safflower seed, cottonseed, canola oils, you will find that high heat, high pressure and strong chemicals and solvents are added to maximize the extraction of the PUFAs contained from within the seed. Being PUFAs, they will go through tremendous amounts of oxidation – in other words, the oils will have become rancid.
Bottled in clear plastic bottles and sitting on market shelves for months only add further light & heat radiation. Finally, you add further damage when you then pour them into your pan to cook with.
This is the untold truth of PUFAs and it’s rather shocking to realize how prevalent they are in our modern diet.
Foods high in saturated fats (SFAs) don’t suffer as much oxidative stress because all their bonds are strong making them much more chemically stable even in the face of heat and light. This makes them ideal for cooking.
What about trans fats?
Trans fats are PUFAs that have been engineered to become a solid in room temperature. They are processed from liquid vegetable oils (PUFAs) where food scientists blast them with hydrogen and under pressure force hydrogen atoms into the kinks. In doing so, they straighten them out in a Frankenstein-like manner that is not found in nature. The result is an unnatural fat that is even more unstable than other PUFAs.
You would do best to stay away from trans fat like the plague.
Here is a simple list of foods that contain trans fat:
- vegetable shortening
- microwavable popcorn
- margarines
- fried fast foods (fried chicken, battered fish, hamburgers, french fries, fried noodles etc…)
- bakery products (muffins, cakes, pastries, doughnuts, etc…)
- non-dairy coffee creamers
- snack junk food (potato chips, cookies, crackers, etc…)
Since it is well known that trans fats are poisonous, some food companies have taken them off their recipes but there are still some who still use them. So it is best to read the food labels in the back of the package (even then, food labelling laws have become a place for loopholes!).
I think that’s enough information for one blog! There was a lot to digest (sorry for the pun!) there but hopefully it has been helpful.
In our next blog, we will go over the importance of omega-6s and 3s, cholesterol and antioxidants. They are all part of this long story on fats!
Summary:
- PUFAs have weak bonds making them chemically reactive or unstable.
- PUFAs that receive heat or light radiation can cause them to oxidize or go rancid.
- Rancid PUFAs can cause various health problems depending on the source of oxidative stress.
- The manufacturing of vegetable oils or the extraction process has already oxidized the PUFAs before you even cook with them.
- Saturated fats are much more stable in heat & light due to its strong chemical structure.
- Trans fats are even more dangerous than PUFAs. STAY AWAY!